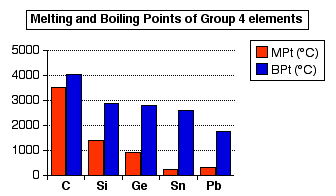
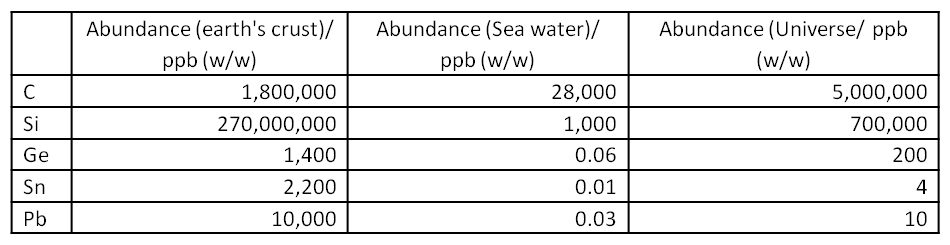
WebElements Periodic Table » Periodicity » Boiling point of highest fluoride » Periodic table gallery
Course: Chemistry, Topic: UNIT 14: TRENDS IN CHEMICAL PROPERTIES OF PERIOD 3 ELEMENTS AND THEIR COMPOUNDS

Silicone oil, for melting point and boiling point apparatuses, Thermo Scientific Chemicals, Quantity: 500 g | Fisher Scientific

SOLVED: For each pair of compounds listed, check the box next to the one with the higher boiling point. compoungt higher boiling point St, Hg SiCl,

SOLVED: Question 30 (1 point) Using the following data, the enthalpy of vaporization for silicon tetrachloride at its determined to be: boiling - point is S1(g) + 2 CI,(g) SiCl (8) 4