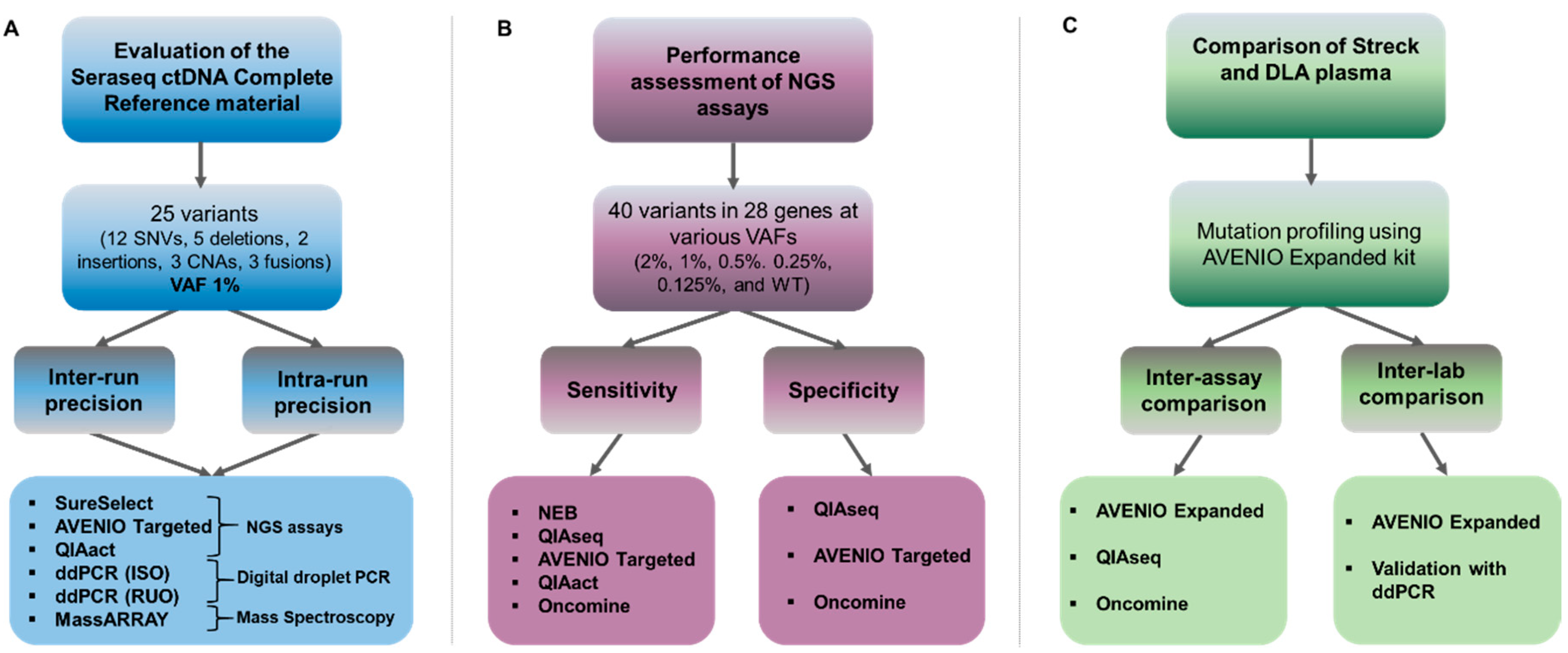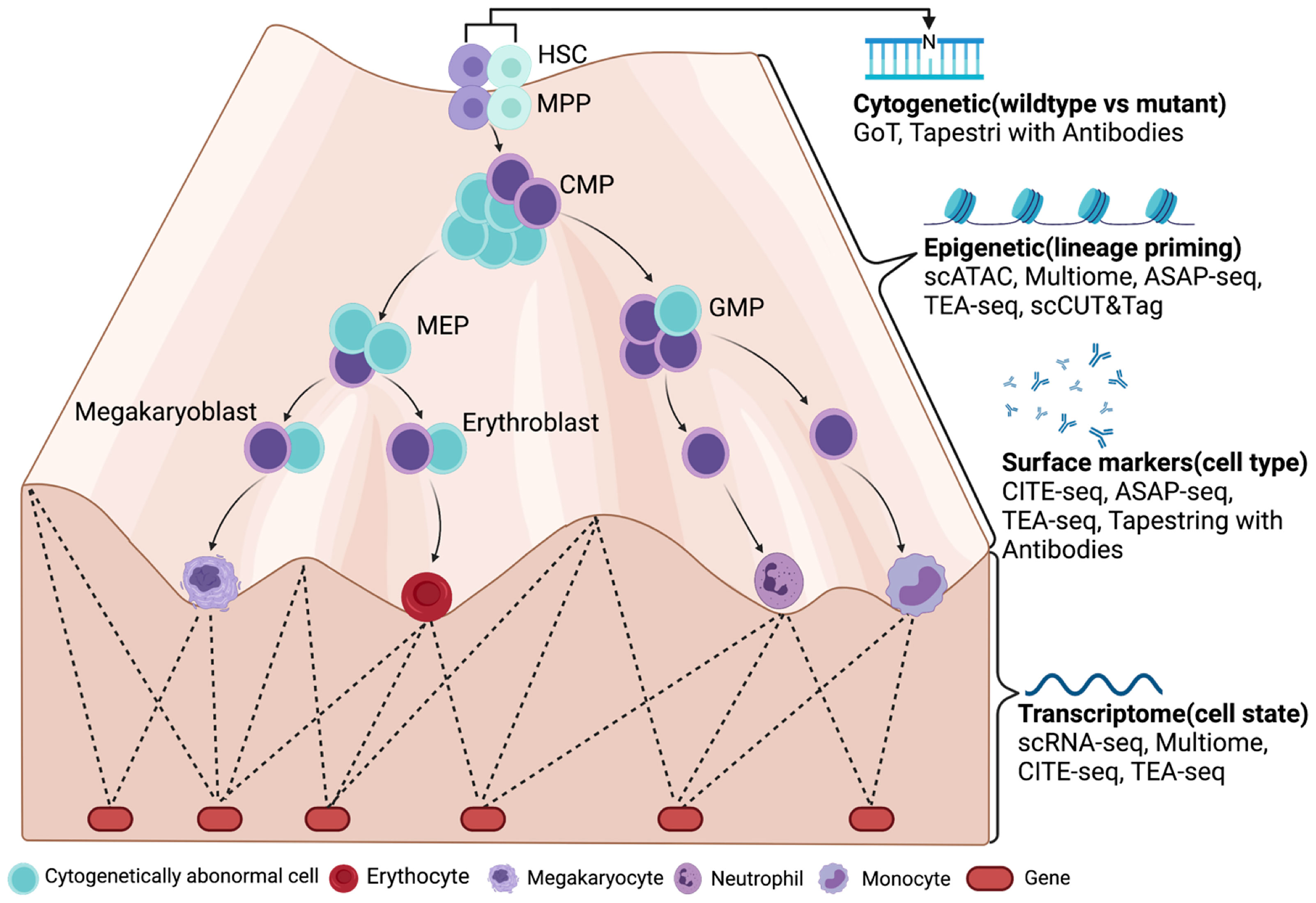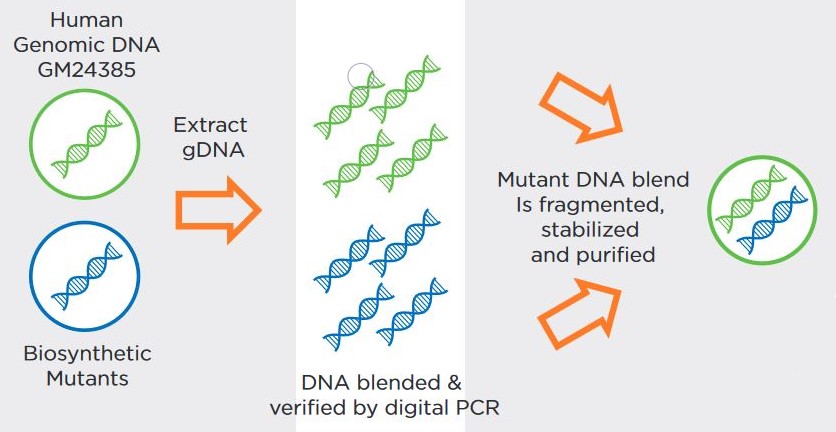
The use of Seraseq® Oncology controls & NGS validation challenges | Webinare | Technical Resources | HiSS Diagnostics
Clinical Validation of a Myeloid Next-Generation Sequencing Panel for Single-Nucleotide Variants, Insertions/Deletions, and Fusi
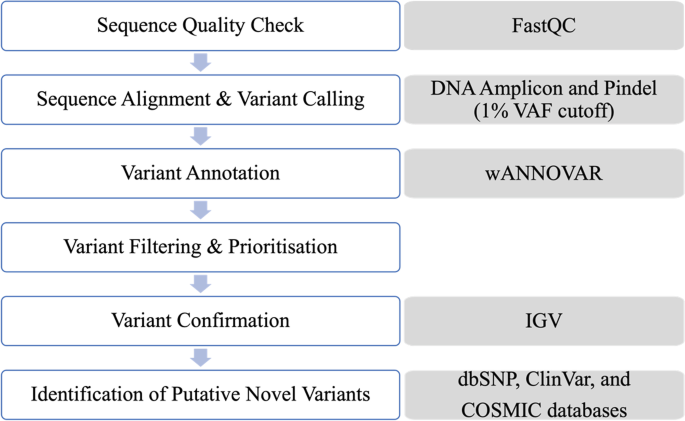
Analysis of genetic variants in myeloproliferative neoplasms using a 22-gene next-generation sequencing panel | BMC Medical Genomics | Full Text