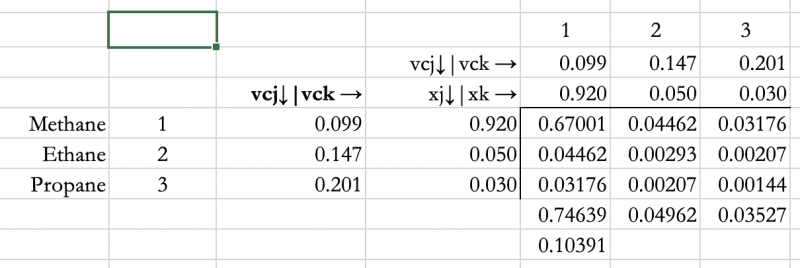
SciELO - Brasil - Modeling the high-pressure behavior of binary mixtures of carbon dioxide+alkanols using an excess free energy mixing rule Modeling the high-pressure behavior of binary mixtures of carbon dioxide+alkanols using

What Is the Optimal Activity Coefficient Model To Be Combined with the translated–consistent Peng–Robinson Equation of State through Advanced Mixing Rules? Cross-Comparison and Grading of the Wilson, UNIQUAC, and NRTL aE Models
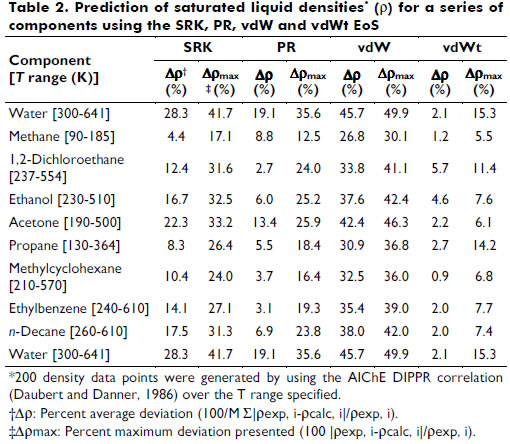
Representation of phase equilibria and densities for complex systems using a van der Waals volume translated equation of state with a UNIFAC mixing rule

Gas-Liquid Equilibrium Prediction of Ternary System (CO<SUB>2</SUB>-Ethanol-Water) at Moderate Pressures and Different Temperatures Using SRK-EOS

Thermodynamic Models for Industrial Applications: From Classical and Advanced Mixing Rules to Association Theories | Wiley

A Systematic Study of Cubic Equations of State with van der Waals Mixing Rules and Different Combining Rules in Predicting the Densities of LNG and Natural Gas Model Systems | Semantic Scholar





![Mixing Rules Equations [15,16] | Download Scientific Diagram Mixing Rules Equations [15,16] | Download Scientific Diagram](https://www.researchgate.net/profile/Arkan-Hadi-2/publication/349615147/figure/tbl1/AS:997184724025353@1614758773903/Mixing-Rules-Equations-15-16_Q320.jpg)